Solids Dissolving In Water Ksp Value
Solubility Product Constants, K sp. Solubility product constants are used to describe saturated solutions of ionic compounds of relatively low solubility.
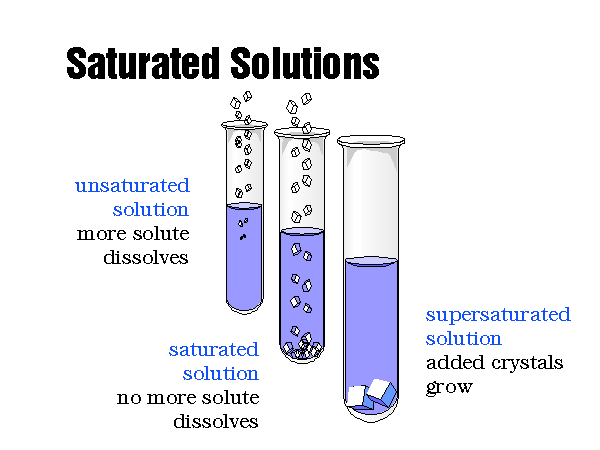

Edit Article wiki How to Determine Solubility. Two Methods: Using Quick Rules Calculating Solubility from the Ksp Community Q&A. Solubility is used in chemistry to


Example #1: Calculate the molar solubility of tin(II) hydroxide in pure water. K sp = 5.45 x 10¯ 27. Solution: 1) Here is the equation for dissociation:
2015–present Senior Instructor II, University of Oregon. 2013–2015 Morrill Professor, Iowa State University. 1998-2013 Professor of Chemistry, Iowa State
Example #2: Aluminum phosphate has a K sp of 9.83 x 10¯ 21. What is its molar solubility in pure water? Solution: 1) Here is the dissociation equation:



The Solubility Product Expression. Silver chloride is so insoluble in water (.0.002 g/L) that a saturated solution contains only about 1.3 x 10-5 moles of AgCl per
Other Aspects of Ionic Equilibria 1 Solubility of Salts – Ksp We now focus on another aqueous equilibrium system, slightly soluble salts. These salts have a

This page contains the name, formula and value of the solubility product of many compounds, as well as the formation constants of selected complex ions.
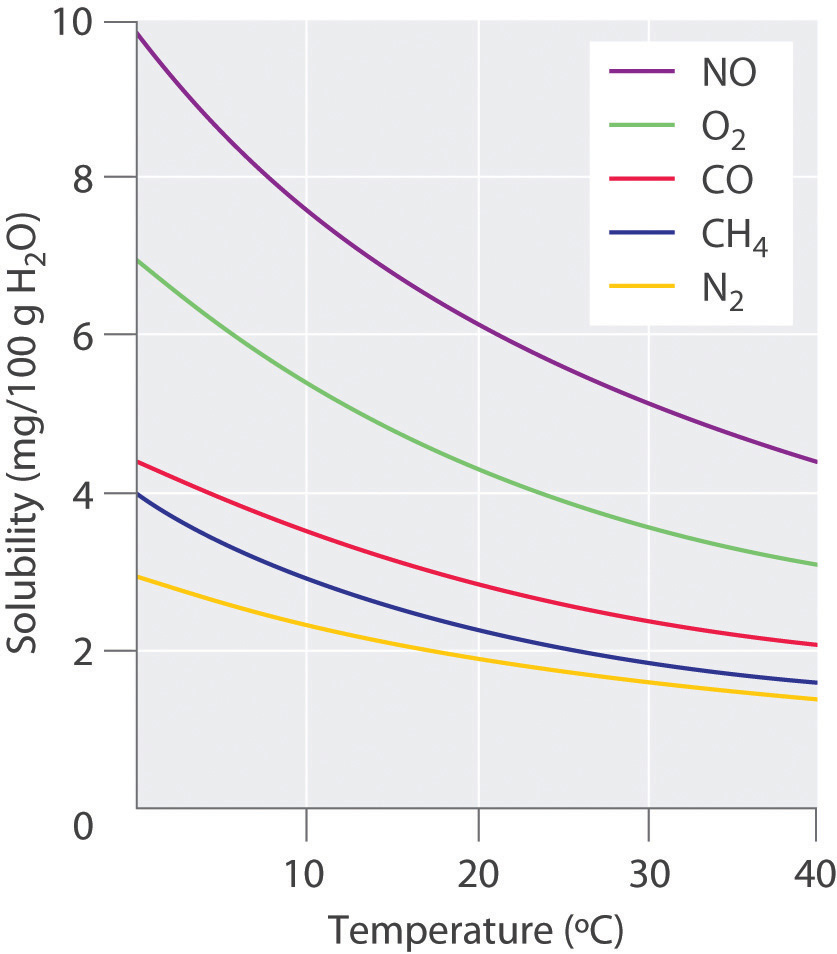
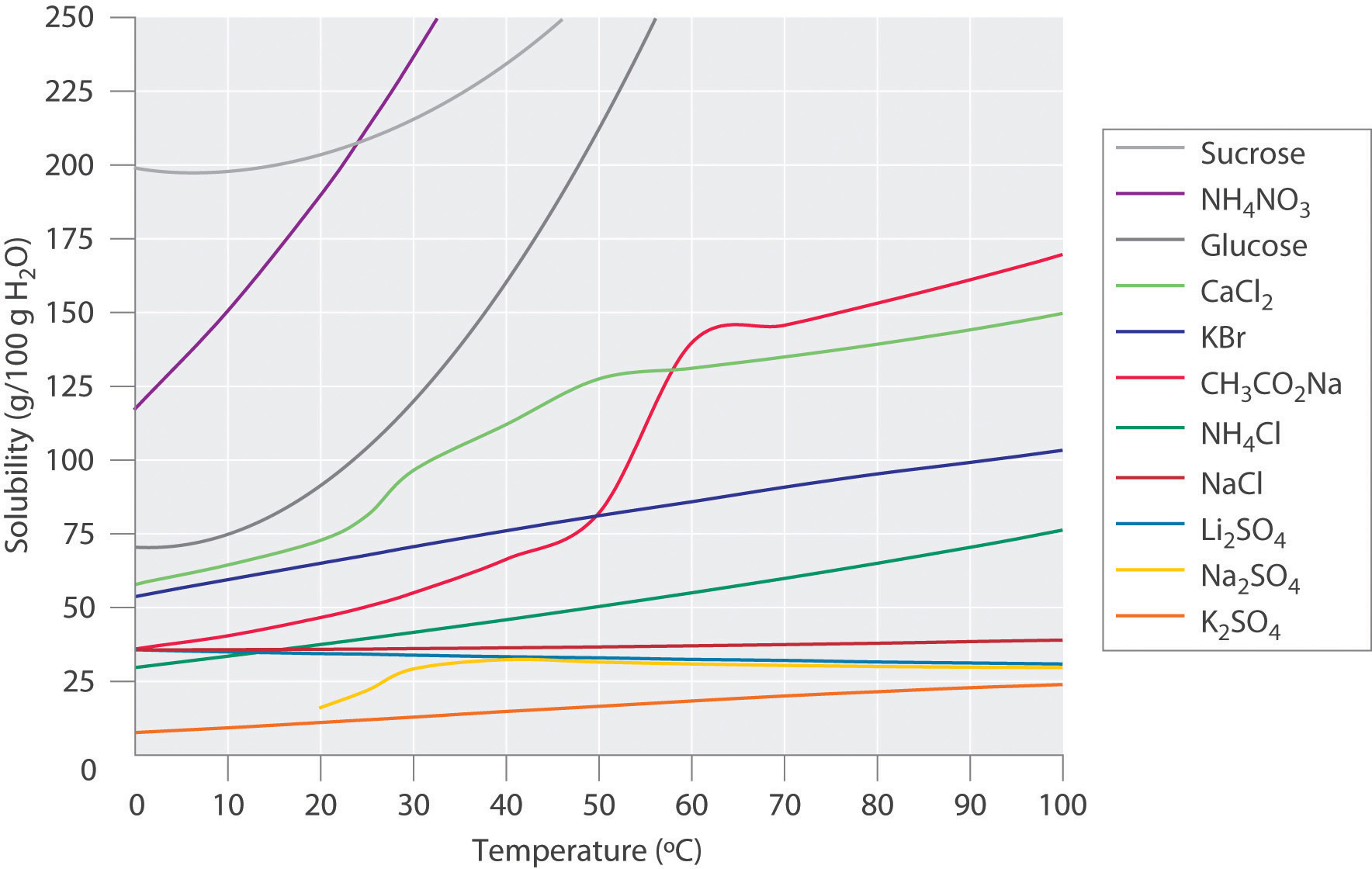
PROPERTIES OF SOLUTIONS . A solution is a mixture of materials, one of which is usually a fluid. A fluid is a material that flows, such as a liquid or a gas.
Water temperature is a physical property expressing how hot or cold water is. Temperature is an important factor to consider when assessing water quality.
Leave a Reply
You must be logged in to post a comment.
No Comments